
Aurion Biotech is pioneering innovative therapies to transform the treatment of blindness and vision loss globally.
Blindness is a global burden that affects millions of patients and their loved ones.
Our lead program addresses blindness caused by Corneal endothelial disease, which affects millions worldwide. Current surgical approaches depend on donor transplantation, limiting access and leaving a significant unmet need.
We are developing therapies designed to broaden availability of treating blindness for both patients and ophthalmologists to address this gap on a global scale.
A platform for regenerative therapies in ophthalmology
Aurion Biotech is advancing a new class of regenerative medicines for the treatment of loss of vision and blindness.
We develop advanced therapeutics that aim to improve patient outcomes, reduce treatment complications, enhance ophthalmologists’ ability to provide care, and optimize utilization of precious donated tissue.
Our therapeutics development platform focuses on manufacturing scale to enable global patient reach, and quality testing to ensure consistent product performance.
Lead program: Corneal Endothelial Cell Therapy (CECT)
Aurion Biotech is advancing Corneal Endothelial Cell Therapy (CECT), a regenerative treatment that aims to replace lost or dysfunctional endothelial cells. Because endothelial cells cannot naturally regenerate, patients experience progressive vision loss. The cells delivered to patients are human corneal endothelial cells obtained from donated corneas. They do not contain any synthetic materials or artificial scaffold. The cells are not obtained by artificial reprogramming of cells that are foreign to the eye. Aurion’s cells are intended to repopulate the endothelium and help re-establish a corneal structure that is virtually indistinguishable from a healthy cornea with healthy fluid balance.
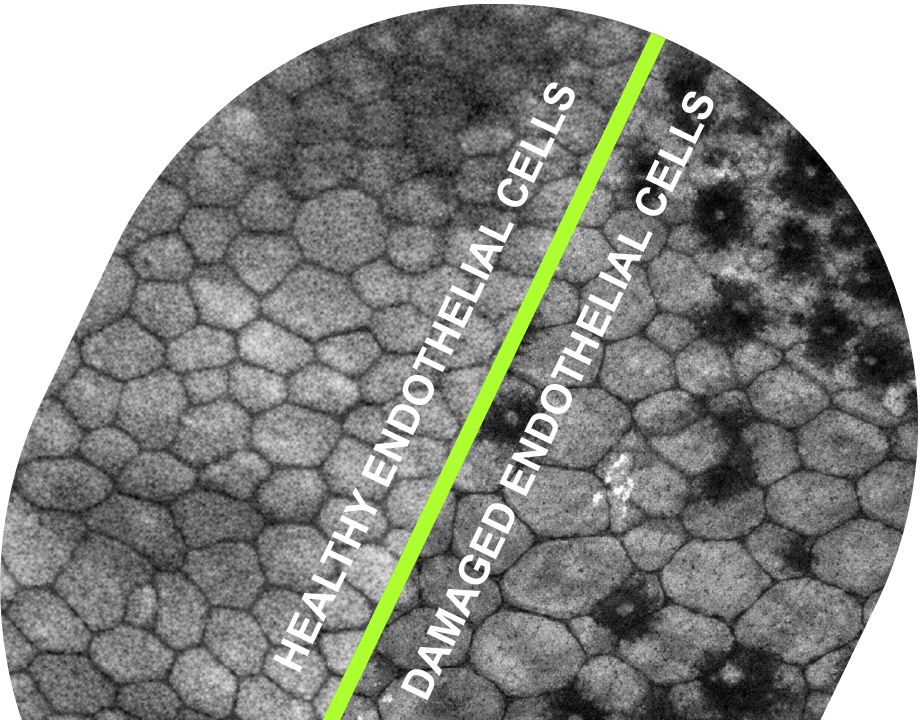
AURN001 is a first-in-class combination therapy made from human, unmodified donor endothelial cells (neltependocel). The cells are delivered in a solution containing a rho-kinase inhibitor compound (Y-27632), which is readily eliminated by the patient’s body after it has helped the cells engraft. AURN001 is designed to restore corneal clarity by recreating a healthy corneal endothelium after diseased cells have been removed.
AURN001 is being developed to:
A platform for regenerative therapies in ophthalmology
Aurion Biotech is advancing a new class of regenerative medicines for the treatment of loss of vision and blindness.
We develop advanced therapeutics that aim to improve patient outcomes, reduce treatment complications, enhance ophthalmologists’ ability to provide care, and optimize utilization of precious donated tissue.
Our therapeutics development platform focuses on manufacturing scale to enable global patient reach, and quality testing to ensure consistent product performance.
Lead program: Corneal Endothelial Cell Therapy (CECT)
Aurion Biotech is advancing Corneal Endothelial Cell Therapy (CECT), a regenerative treatment that aims to replace lost or dysfunctional endothelial cells. Because endothelial cells cannot naturally regenerate, patients experience progressive vision loss. The cells delivered to patients are human corneal endothelial cells obtained from donated corneas. They do not contain any synthetic materials or artificial scaffold. The cells are not obtained by artificial reprogramming of cells that are foreign to the eye. Aurion’s cells are intended to repopulate the endothelium and help re-establish a corneal structure that is virtually indistinguishable from a healthy cornea with healthy fluid balance.
AURN001 is a first-in-class combination therapy made from human, unmodified donor endothelial cells (neltependocel). The cells are delivered in a solution containing a rho-kinase inhibitor compound (Y-27632), which is readily eliminated by the patient’s body after it has helped the cells engraft. AURN001 is designed to restore corneal clarity by recreating a healthy corneal endothelium after diseased cells have been removed.
AURN001 is being developed to:

Aurion Biotech collaborates with clinicians, researchers, and partners committed to advancing the future of corneal treatments. Get in touch to learn more about our science, programs, and global mission